CAR T-Cell Therapy: Is it the New Way to Fight ALS?
- websitethestembull
- 19 hours ago
- 5 min read
Written by Parthasarathe Jayashanker
Published by Nur Sofia Balqis
Throughout our daily lives, most of us take control of our bodies for granted—until we lose it. Imagine noticing a twitch in your hand, then struggling to walk, and finally finding it extremely tough to breathe. Welcome to the world of ALS, or amyotrophic lateral sclerosis. It’s a progressive disease where motor neurons, the cells that help your brain talk to your muscles, start dying off. Once those neurons fade, muscles stop working, but your mind stays sharp. It’s rare, but globally, ALS makes a big impact: an estimated over 300,000 people are living with it, and after symptoms show up, most only make it another two to five years. While outliers like Professor Stephen Hawking survived for decades, his case was exceptional; diagnosed at 21, he had a rare, slow-progressing form of the disease that spared his respiratory muscles far longer than the typical aggressive progression seen in older patients, combined with elite 24-hour care that managed respiratory complications. How about the treatments we have right now? Unfortunately, they only provide minimal aid. Survival improves, but not by much.
Figure 1: Professor Stephen Hawking, who lived with ALS for over five decades, remains the most prominent example of the disease’s impact on motor neurones.

ALS: What’s Really Going On?
ALS, also known as Lou Gehrig’s disease (named after the legendary baseball player who lived with it), basically breaks the pathway from your brain to your muscles. Over time, this means weakness slowly creeps in, and speech or swallowing becomes difficult. Sure, it’s all about those motor neurons dying, but lately researchers have been paying more attention to microglia, the brain’s immune cells. Usually, microglia clear junk and keep nerves healthy, but in ALS, they go into overdrive. Microglia can shift from protective to pro-inflammatory states by releasing cytokines like TNF-α, which actually makes things worse for neurons. Doctors will diagnose ALS by examining patients using EMG (electromyography), and sometimes by doing genetic testing, especially when the disease runs in families.
Figure 2: The Neuroinflammatory Pathway

Source: Adapted from Carata et al. (2023). Created with Biorender.com
What’s CAR T-Cell Therapy Anyway?
CAR T-cell therapy is a form of immunotherapy, where it’s all about using our own immune system in a high-tech way. Here’s how it works: scientists start off by taking your T-cells (a type of immune cell) and rewire them genetically to spot and attack trouble spots. These cells are engineered to use something called a chimeric antigen receptor (CAR) to hunt specific targets and trigger a precise immune response (Katsaros DE, 2025). This therapy’s already made huge waves in fighting blood cancers, where it’s been surprisingly effective.
Figure 3: A step-by-step breakdown of the process of CAR T-Cell Therapy
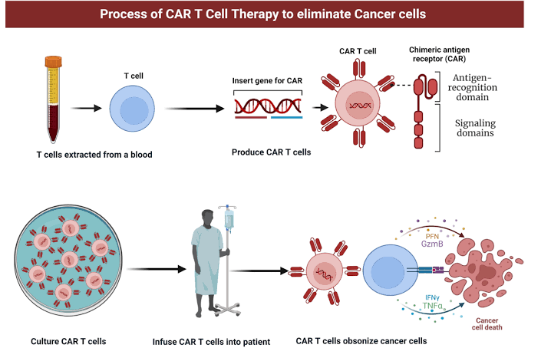
Can CAR T-Cell Therapy Tackle ALS?
Researchers are now testing if CAR T-cell therapy works for ALS and other brain diseases. Recent experimental studies have explored CAR T-cells that focus on a protein called uPAR, which shows up in activated microglia stirring up inflammation (New Scientist, 2026).
In animal studies, these engineered cells managed to:
Get into the central nervous system
Seek out and destroy activated microglia
Slash neuroinflammation by about 70%
Protect nerve cells and help animals live longer
Those results hint that this therapy might tame bad inflammation in the brain without harming good tissue (New Scientist, 2026). But all of this is still happening in the lab - human trials are yet to come.
How Does It Work? From Mice to Early Human Research
In mouse models of ALS, CAR T-cells that target uPAR turned down inflammation and slowed the disease. Mice kept their muscle function longer compared to others treated with standard drugs like riluzole (New Scientist, 2026). The same immune-engineering tricks have helped reduce brain plaques in other diseases, too, so there could be wider potential here (Washington University School of Medicine, 2026). Early clinical research is just starting to push CAR T-cell therapy beyond cancer. Schools like UCSF are developing CAR T-cell options, but for now, most trials stick to oncology and brain tumors, not ALS yet (UCSF Clinical Trials, 2026). Biotech firms like Allogene Therapeutics are working on “off-the-shelf” versions, hoping to make treatment faster and cheaper - though again, their focus is cancer for now (Allogene Therapeutics, 2026).
Figure 4: Mouse models are instrumental in testing before human trials begin.

Safety: What We’ve Learned from Cancer
CAR T-cell therapy works well, but it’s not risk -free. One nasty problem is cytokine release syndrome (CRS), where the immune system can go wild, causing dangerous body-wide inflammation. There’s also risk of neurotoxicity and other immune complications. That’s why scientists are careful. The good news: early animal studies in ALS suggest that targeting uPAR in microglia could make the immune response more controlled and local, with fewer side effects. Still, we need trials in actual people to know for sure.
Ethics & Accessibility: Big Challenges Ahead
Right now, CAR T-cell therapy isn’t cheap. For cancer, the costs often exceed $400,000 per patient because the process is so precise and personal, although emerging centres in countries such as India have begun offering it at significantly lower costs, ranging from approximately $3o,ooo to $85,000. But it’s not all good news; now we have to wrestle with tough choices about who actually gets access to these kinds of treatments and who’s left waiting. To fix this, researchers are developing universal, donor-derived CAR T-cells, so “off-the-shelf” options might cut costs and make access fairer. Expanding out more global clinical trials can also help to ensure more equitable patient representation (Allogene Therapeutics, 2026).
Figure 5: A global comparison of current costs of CAR T-Cell Therapy, highlighting the significant barriers to patient accessibility.

Source: Data provided by IndiCure Health Tours (2025).
Looking Forward
CAR T -cell therapy started off as a cancer breakthrough, but it’s quickly shifting toward brain diseases like ALS. Even though the current evidence is still largely preclinical, early studies are very promising. Tweaking the immune system might actually change the course of ALS, and honestly, that could be a game changer, since older treatments haven’t delivered much hope. As science moves forward and research advances, this therapy signals a bigger push toward precision medicine, where treatments are designed in a way not only to manage symptoms, but to directly address the underlying mechanisms of the disease. That’s a big deal for ALS and maybe for other stubborn diseases, too.
References
UCSF Clinical Trials, 2026. https://clinicaltrials.ucsf.edu/trial/NCT07081646
Katsaros DE, Mougiakakos D. CAR T-cell therapy for neurological disorders: scientific rationale and mechanistic insights. Ther Adv Neurol Disord. 2025 Dec 10;18:17562864251396039. doi: 10.1177 /17562864251396039. PMID: 41394288; PMCID: PMC12696306.
Wegorzewska M. Engineered immune cells help reduce toxic proteins in the brain Washington University School of Medicine, 2026. https://medicine.washu.edu/news/engineered-immune-cells-help-reduce-toxic-proteins-in-the-brain/
Carata, E., Muci, M., Di Giulio, S., Mariano, S., & Panzarini, E. (2023). Looking to the future of the role of macrophages and extracellular vesicles in neuroinflammation in ALS. International Journal of Molecular Sciences, 24(14), 11251. https://doi.org/10.3390/ijms241411251
Aditi. (2025, July 19). CAR T-cell therapy in India: Affordable cancer treatment. IndiCure Health Tours. https://www.indicure.com/blog/cancer-treatment-with-car-t-cell-therapy-in-India/
Comments