Can we use viruses to treat cancer?
- websitethestembull
- 19 hours ago
- 6 min read
Written by Ashley Ong
Published by Nur Sofia Balqis
Most people see viruses as our enemies. From the influenza virus causing the flu, to the varicella-zoster virus giving us chicken-pox, to Covid-19 which caused a global lockdown, it seems like they do nothing but cause trouble. But what if getting infected by a virus could actually save your life?
Cancer incidence is rising globally. Granted, as treatment for other diseases improves, and people live longer lives, it is expected that we will see an increase in cases. After all, the biggest risk factor for cancer is simply getting older!
The worrying trend is the increase in cancer cases in young people, or in other words, early-onset cancer. There are multiple theories for what might be causing this, ranging from obesity and alcohol consumption, to overabundance of microplastics in our bodies and environmental pollution, to imbalances in the gut microbiome. While a definitive cause is unclear (it’s likely these are all contributing factors), what is clear is that cancer is going to become more common, with the global incidence of early-onset cancer increasing by 79.1% between 1990 and 2019 (Zhao et al., 2023).
This highlights the need for more tailored and sophisticated cancer therapies to fight more effectively against the disease. One such emerging treatment is oncolytic virus therapy (or OVT, for short). A type of immunotherapy, oncolytic viruses have evolved and/or are genetically engineered to selectively infect cancer cells while sparing healthy cells, thus providing a more specific and targeted response compared to traditional treatment methods like chemotherapy.
So how exactly does it work?
Oncolytic viruses kill cancer cells in two ways, the first being direct oncolysis (the lysis/breakdown of cancer cells).
Tumors are natural hotspots for viral replication due to the impaired anti-viral defenses and rapid replication of cancer cells, and viruses can be engineered to be even more tumor-specific.
As with infection of healthy cells, the viruses first bind to receptors on the cell surface membrane of the cell, tricking the cell into taking the virus in.
Once inside, its genetic material is released.
The viral genetic code is read and transcribed to make viral proteins.
Once enough viral components have been produced, self assembly of new virions occurs.
These new virions burst out of the cell, killing it in the process.
The second mode of action is activation of the immune system. The lysis of the cancer cell causes all of the cell contents to spill out into the surrounding tissue, besides causing inflammation. When immune cells rush to the site as a result of this inflammation, antigen-presenting cells pick up both the viral antigens and the tumor-associated antigens (which the cancer may have previously been hiding). These are presented to T-cells in the lymph nodes. Crucially, this not only activates T-cells that recognise the virus, but also T-cells that recognise the tumor antigens. This means that both virus-infected cells and uninfected cancer cells are targeted and killed by the immune system. Thus, a systemic anti-tumor response is triggered, potentially causing the destruction of other distant, untreated tumors in the body (the abscopal effect), as seen with the T-VEC oncolytic immunotherapy. Moreover, some of these T-cells differentiate into memory-T cells, acting as a personalised cancer vaccine which kills the cancer cells if they come back again!

Figure 1: mechanism of oncolytic virus therapy
Why is it useful?
As with all immunotherapies, a major part of oncolytic virus therapy is its stimulation of the body’s immune system to fight cancer. But why wasn’t the immune system working against cancer in the first place?
We all know that our immune systems are great at destroying foreign invaders - a common cold, for example, doesn’t tend to last more than a week. But this isn’t the case with cancer. While our immune system does often destroy pre-cancerous cells before a tumor is formed, cancer is excellent at immune evasion, for a few reasons.
They can “tell” immune cells not to attack them: cancer cells often express ligands such as PD-L1 and PD-L2 on their surfaces, which bind to the complementary receptors on T-cells to deactivate them. In addition, they secrete immunosuppressive chemicals which prevent the growth and activation of tumor-attacking immune cells, while promoting the development of immunosuppressive cells.
They can “hide their identity”: they downregulate the production of antigens that identify them as tumor cells, making it harder for T-cells to recognise them.
They change the environment: the TME or tumor-microenvironment is often hypoxic and low in nutrients, which suppresses the adaptive immune system.
They recruit immunosuppressive cells, for example T-regulatory cells, which inhibit the anti-tumor response.
As such, oncolytic virus therapy is so useful because it can convert a “cold” (immunosuppressive) tumor into a “hot” one, by essentially exposing the hiding cancer and alerting the immune system of its existence.
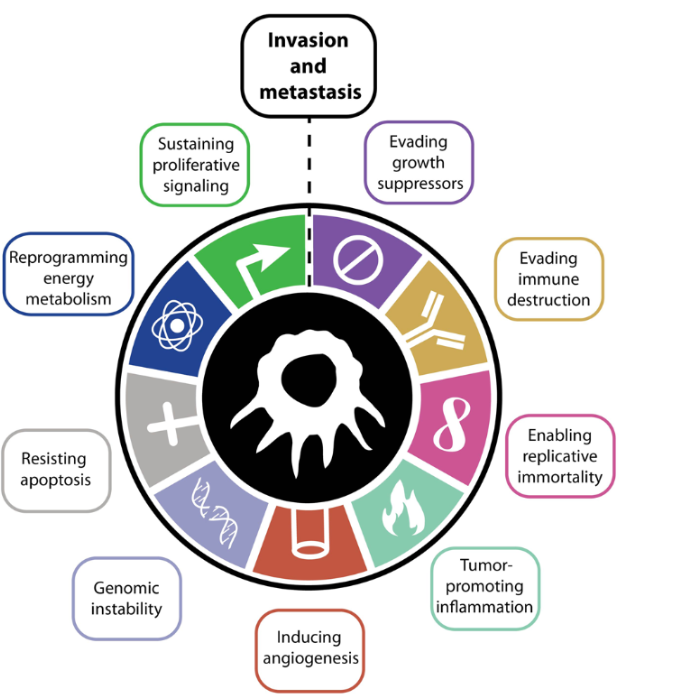
Figure 2: The 10 hallmarks of cancer
Limitations
This all sounds good in theory - but the reality isn’t that simple. Remember what I said earlier about the activation of T-cells that recognise the virus? Unfortunately, this means that the oncolytic virus is quickly removed from circulation, so the timeframe of direct oncolysis is limited. Nevertheless, this is generally not a huge issue as the virus would’ve had time to alert and activate the immune system before being cleared off. However, in some cases, patients have pre-existing immunity to an OV (e.g. the Vaccinia virus, which originates from the smallpox vaccine) - meaning the virus wouldn’t have a chance to lyse cancer cells at all.
Furthermore, a hallmark of cancer is genomic instability - in other words, cancer is constantly mutating. As a result, surface antigens are always changing and may differ from site-to-site, or even from cell-to-cell, making widespread viral infiltration challenging. This is also why a systemic anti-tumor response isn’t always achieved.
The excellent immune-evading capabilities of cancer, from the tumor-microenvironment to the expression of immune-inhibiting ligands, also means that the immune response started by the virus can be stifled.
In addition to this, physically administering the virus isn’t easy either. The two main methods currently are intratumoral injection and intravenous infusion.
Intratumoral injection delivers the viral particles directly to the tumor, reducing the risk of the virus being cleared by the immune system before it has a chance to work. However, this can be challenging for visceral tumors, may cause bleeding, and is unsuitable for metastatic cancers.
Intravenous infusion is better at targeting metastatic or difficult-to-reach cancers as the virus travels in the bloodstream, however this also increases the risk of antibodies clearing the virus before it can cause oncolysis.
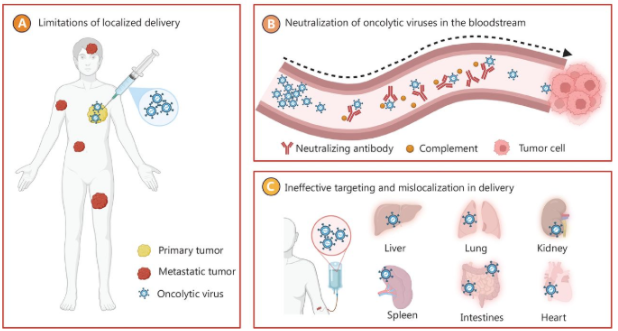
Figure 3: complications of OVT administration
Future directions
Thankfully, scientists are researching lots of ways to mitigate these challenges and improve the efficacy of virotherapy.
To streamline the delivery of OVTs, and minimise immune-clearance, cell-based and bionanocarrier strategies are being devised. These act as carriers to transport the virus to the target site, and have already had some clinical success. For example, the ONCOTECH delivery system (Peng, et. al 2025), which attaches oncolytic viruses to T-cells, protects OVs from being neutralised by the immune system and improves their tumor-targeting abilities.

CRISPR-Cas9 and other synthetic biology techniques are also being used to aid immune evasion (e.g. by changing the viral surface antigens), while also improving anti-tumor abilities (e.g. by modifying them to produce cytokines).
Developments in genetic and immune profiling technologies are also enabling clinicians to customise therapy to suit the unique tumor profile of each patient, leading to more successful outcomes and helping them determine if OVT monotherapy or combination therapy would be more appropriate.
In short, the future for oncolytic virus therapy is bright. While this method of treatment still faces many limitations, the next few years of research could significantly improve its efficacy, paving the way for more effective treatments for a devastating disease predicted to continue to rise. Who would’ve thought that one of our worst biological enemies might become one of our closest allies!
References:
Beer, G. (2019, February 28). Why doesn’t the immune system attack cancer cells? Science surgery: - Cancer Research UK - Cancer news. Cancer Research UK - Science Blog. https://news.cancerresearchuk.org/2019/02/28/science-surgery-why-doesnt-the-immune-system-attack-cancer-cells/
Jones, G. (2015, February 4). Why are cancer rates increasing? - Cancer Research UK - Cancer news. Cancer Research UK - Science Blog. https://news.cancerresearchuk.org/2015/02/04/why-are-cancer-rates-increasing/
Maroun, J., Muñoz-Alía, M., Ammayappan, A., Schulze, A., Peng, K.-W., & Russell, S. (2017). Designing and building oncolytic viruses. Future Virology, 12(4), 193–213. https://doi.org/10.2217/fvl-2016-0129
Rohaan, M. W., Stahlie, E. H. A., Franke, V., Zijlker, L. P., Wilgenhof, S., van der Noort, V., van Akkooi, A. C. J., & Haanen, J. B. A. G. (2022). Neoadjuvant nivolumab + T-VEC combination therapy for resectable early stage or metastatic (IIIB-IVM1a) melanoma with injectable disease: study protocol of the NIVEC trial. BMC Cancer, 22(1). https://doi.org/10.1186/s12885-022-09896-4
Tufail, M., Jiang, C.-H., & Li, N. (2025). Immune evasion in cancer: mechanisms and cutting-edge therapeutic approaches. Signal Transduction and Targeted Therapy, 10(1). https://doi.org/10.1038/s41392-025-02280-1
Xiao, D., Zhang, H., Liu, Y., Li, Y., Li, G., & Ning, Y. (2026). Oncolytic viruses: advanced strategies in cancer therapy. Signal Transduction and Targeted Therapy, 11(1). https://doi.org/10.1038/s41392-025-02343-3
Zhao, J., Xu, L., Sun, J., Song, M., Wang, L., Yuan, S., Zhu, Y., Wan, Z., Larsson, S., Tsilidis, K., Dunlop, M., Campbell, H., Rudan, I., Song, P., Theodoratou, E., Ding, K., & Li, X. (2023). Global trends in incidence, death, burden and risk factors of early-onset cancer from 1990 to 2019. BMJ Oncology, 2(1), e000049. https://doi.org/10.1136/bmjonc-2023-000049
Figure 1:
Oncolytic Virus Platforms | Vyriad. (2024, May 3). Vyriad. https://vyriad.com/science/oncolytic-virus-platforms/
Figure 2:
Meirson, T., Gil-Henn, H., & Samson, A. O. (2019). Invasion and metastasis: the elusive hallmark of cancer. Oncogene, 39(9), 2024–2026. https://doi.org/10.1038/s41388-019-1110-1
Figures 3 and 4:
Peng, Z., Kalim, M., & Lu, Y. (2025). Improving systemic delivery of oncolytic virus by cellular carriers. Cancer Biology & Medicine, 21(12), 1104–1119. https://doi.org/10.20892/j.issn.2095-3941.2024.0390

Comments